Trusted
Solutions for
Calcium Sennosides
Rooted in Tradition & Driven by Science
Trusted Solutions for Calcium Sennosides
Trusted for Gentle & Effective Relief
Calcium Sennosides & Senna Extracts
Our commitment to harnessing potential herbal extracts is reflected in each product line of KP Phyto Extracts. Amongst the same, the one which continues to carry the banner is Calcium Sennoside, A well-established, natural aid for digestive health.
We offer quality extracts of Calcium sennoside and Senna Extracts with our decades of experience in manufacturing and supplying. Our focus remains on consistency, compliance and quality; the same values that have shaped us from the very beginning. We take pride in being a trusted partner for those seeking dependable, plant-based solutions
The Essence of Senna
Our Natural Source
Calcium Sennoside is extracted from the leaves of Cassia angustifolia, commonly known as Senna. This remarkable herb, native to Sudan and Arabia, has found a natural home in the Indian agro-climatic conditions, thriving in regions like Rajasthan, southern India, Gujarat, and Kutch in western India, where it is cultivated.
Senna leaves are known for their distinct morphology: an erect, smooth stem with spreading branches, bearing characteristic leaflets. Premium quality Senna is recognized by its bright, yellowish-green color, a faint, peculiar aroma reminiscent of green tea, and a mucilaginous, sweetish, slightly bitter taste.

Our Calcium Sennosides Product Range
We are committed to providing Calcium Sennoside in various grades to suit your specific applications and quality standards:
Calcium Sennosides A&B
Available in 20%, 40%, and 60% concentrations.
Adhering to USP Grade standards.
Calcium Sennosides A&B
Also offered in 20%, 40%, and 60% concentrations.
Conforming to B.P. Grade standards.
Calcium Sennosides A&B
Available in 20%, 40%, and 60% concentrations.
Verified by HPLC Method.
Sennosides
An 80% concentration.
As per USP Grade.
Sennosides Leaf Powder
Contains more than 2% of Sennoside.
Raw Senna Herb
Available as such and in powder form, including leaves of different size grades, pods, and stems.
Pharmacology
The Senna plant is a natural source of calcium sennoside, a substance known for its mild laxative effects. For centuries, traditional medicine and contemporary health formulations have utilized sennosides, the active anthraquinone derivatives found in senna leaves. Even though it is mild, the laxative property works well enough to safely and consistently treat occasional constipation.Therefore, Calcium Sennoside functions as a mild purgative by directly affecting intestinal walls, which encourages the colon’s peristaltic motions and facilitates smooth bowels. This makes it a very adaptable component for creating a tried-and-true, safe dietary supplement.
Senna is also a common ingredient in weight-loss products like “dieter’s teas” and a major ingredient in a number of commercial stimulant laxatives. By limiting calorie absorption, these formulations frequently take advantage of their dual effects of decreasing appetite and speeding up digestive transit, which may aid in temporary weight loss. Notably, the active ingredients carry out their purgative actions without causing a substantial change in metabolism, and the laxative effect is further amplified when mixed with bitter herbs.

Clinically Proven Efficacy
How Calcium Sennosides Works
For centuries, Sennosides, the active anthraquinone derivatives found in Senna leaves, have been traditionally and modernly used as a proven, natural remedy for occasional constipation. Calcium Sennoside possesses excellent natural laxative properties, making it a very flexible and effective ingredient for safe formulations.
Natural Laxative
Senna acts as a soft purgative due to its rich Anthraquinone content, stimulating colon activity by increasing peristaltic movements.
Effective in Weight Management
Commonly used in “dieter’s teas” and laxatives, Senna helps suppress appetite and accelerates digestion, supporting rapid weight loss.
Active and Potent
Its compounds remain unchanged through digestion, ensuring consistent purgative action throughout the system.
Enhanced Efficacy with Bitters
The laxative effect is further intensifies when used in combination with pure bitters.
Natural Laxative
Senna acts as a soft purgative due to its rich Anthraquinone content, stimulating colon activity by increasing peristaltic movements.
Effective in Weight Management
Commonly used in “dieter’s teas” and laxatives, Senna helps suppress appetite and accelerates digestion, supporting rapid weight loss.
Active and Potent
Its compounds remain unchanged through digestion, ensuring consistent purgative action throughout the system.
Enhanced Efficacy with Bitters
The laxative effect is further intensified when used in combination with pure bitters.
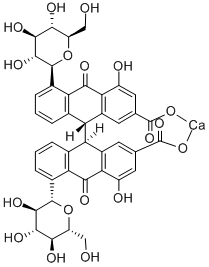
Principle Constituents
Key Actives
Contains Anthraquinone derivatives and their glucosides, known as Sennosides.
Primary Glycosides
Includes compounds like Rhein, Rhein 8-glucoside, and Rhein 8-diglucoside.
Flavonoid Components
Features 8-glucosi kaempferol, aloe-emodin, and Isorhamnetin (free and glucoside forms).
Additional Compounds
Also contains myricyl alcohol and Chrysophanic Acid.
Mineral Content
Senna ash is rich in earthy and ashy carbonates.
Phytochemical Richness
A potent blend of bioactive compounds contributing to its therapeutic properties.
Reach out to the experts at K Patel Phyto.
Looking for an ingredient solution for your specific application

